Plastic Injection Molding products1 often require surface modification to enhance their aesthetic appeal, durability, or conductivity. The two primary methods for surface treatment are coating et plating.
However, plastics present unique challenges:
- Low Surface Energy: Most plastics have high crystallinity and low polarity, making adhesion difficult.
- Insulation: Plastics are non-conductive, preventing direct electroplating.
Therefore, rigorous pretreatment is essential to improve adhesion for coatings and to create a conductive base layer2 for plating.

1. Pretreatment for Coating Processes
The goal of pretreatment for coatings is to remove contaminants and increase surface energy (wettability) to ensure the paint or coating adheres firmly.
1.1 Degreasing (Cleaning) Similar to metal treatment, the first step is removing oil, grease, and mold release agents.
- Organic Solvents: Used for removing paraffin, beeswax, and organic dirt. The solvent must not dissolve or swell the plastic. It should be volatile, non-toxic, and non-flammable.
- Alkaline Solutions: Suitable for alkali-resistant plastics. These solutions contain caustic soda, alkaline salts, and surfactants (e.g., OP series alkylphenol polyoxyethylene ether) which are low-foaming and leave no residue.
1.2 Surface Activation Activation introduces polar groups or roughens the surface to increase surface energy. Common methods include chemical oxidation, flame oxidation, solvent vapor etching, and corona discharge.
Chemical Oxidation (Chromic Acid Treatment) This is the most widely used method. A typical formulation involves a mixture of Potassium Dichromate, Water, and Concentrated Sulfuric Acid.
| Component | Percentage / Concentration |
|---|---|
| Potassium Dichromate | 4.5% |
| Water | 8.0% |
| Concentrated Sulfuric Acid (>96%) | 87.5% |
ABS Plastic Treatment: For high-quality coatings, ABS plastics can be etched after degreasing using a dilute chromic acid solution (Chromic acid 420g/L, Sulfuric acid 200ml/L) at 65°C–70°C for 5–10 minutes.
Note : While chromic acid etching provides uniform treatment even on complex shapes, it poses safety hazards and environmental pollution risks.

2. Pretreatment for Plating Processes
The objective here is twofold: improve adhesion and create a conductive metal(base layer) on the insulating plastic.
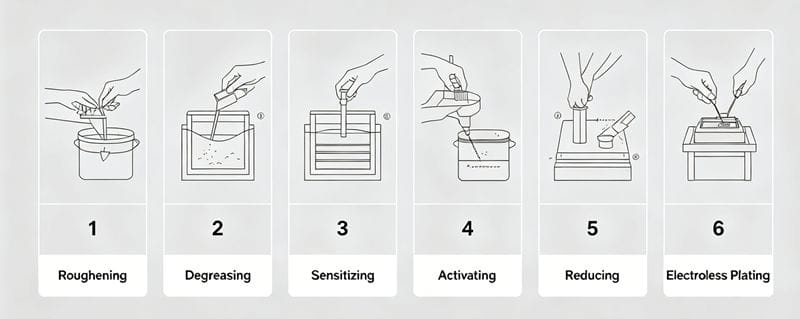
The process flow is: Mechanical/Chemical Roughing → Degreasing → Sensitization → Activation → Reduction → Electroless Plating
2.1 Roughening and Degreasing
- Mechanical vs. Chemical Roughening: Roughening increases the contact area. Mechanical roughening is generally less effective, providing only about 10% of the adhesion strength achieved by chemical roughening.
- Degreasing: Follows the same principles as the coating process described above.
2.2 Sensitization This step involves adsorbing easily oxidizable substances (like Stannous Chloride ou Titanium Trichloride) onto the plastic surface. This prepares the surface for the subsequent activation step.
2.3 Activation The sensitized surface is immersed in a solution containing noble metal salts (oxidizing agents). The noble metal ions are reduced to metallic colloids (catalytic nuclei) which deposit on the surface. These nuclei act as catalysts for the plating reaction.
2.4 Reduction and Electroless Plating
- Reduction Treatment: Before plating, the part is dipped in a reducing agent (e.g., Formaldehyde for copper plating, Sodium Hypophosphite for nickel plating) to remove residual activator.
- Electroless Plating: This is the critical step. A thin layer of metal is chemically deposited onto the plastic, making it conductive. Once conductive, standard electroplating can proceed.
Summary of Process Differences
[Coating vs. Plating Pretreatment]
| Feature | Coating Pretreatment | Plating Pretreatment |
|---|---|---|
| Primary Goal | Improve wettability and adhesion | Create a conductive metal layer |
| Key Step | Surface Activation (Oxidation) | Electroless Plating (Metallization) |
| Conductivity | Not required | Essentiel (must be achieved before plating) |
| Complexity | Modéré | High (Multiple chemical steps) |




